Smallpox was an
infectious disease caused by either of two
virus variants,
Variola major and
Variola minor.
[1] The disease is also known by the
Latin names
Variola or
Variola vera, derived from
varius ("spotted") or
varus ("pimple"). The disease was originally known in English as the "
pox"
[2] or "
red plague";
[3] the term "smallpox" was first used in Britain in the 15th century to distinguish variola from the "great pox" (
syphilis).
[4] The last naturally occurring case of smallpox (
Variola minor) was diagnosed on 26 October 1977.
[5]
Smallpox localized in small
blood vessels of the skin and in the mouth and throat. In the skin it resulted in a characteristic
maculopapular rash and, later, raised fluid-filled
blisters.
V. major produces a more serious disease and has an overall
mortality rate of 30–35%.
V. minor causes a milder form of disease (also known as
alastrim,
cottonpox,
milkpox,
whitepox, and
Cuban itch) which kills about 1% of its victims.
[6][7] Long-term complications of
V. major infection include characteristic scars, commonly on the face, which occur in 65–85% of survivors.
[8] Blindness resulting from
corneal ulceration and scarring, and limb deformities due to arthritis and
osteomyelitis are less common complications, seen in about 2–5% of cases.
Smallpox is believed to have emerged in
human populations about 10,000 BC.
[4] The earliest physical evidence of it is probably the pustular rash on the mummified body of Pharaoh
Ramses V of Egypt.
[9]
The disease killed an estimated 400,000 Europeans annually during the
closing years of the 18th century (including five reigning
monarchs),
[10] and was responsible for a third of all blindness.
[6][11] Of all those infected, 20–60%—and over 80% of infected children—died from the disease.
[12] Smallpox was responsible for an estimated 300–500 million deaths during the 20th century.
[13][14][15] As recently as 1967, the
World Health Organization (WHO) estimated that 15 million people contracted the disease and that two million died in that year.
[5]
After
vaccination campaigns throughout the 19th and 20th centuries, the WHO certified the eradication of smallpox in 1979.
[5] Smallpox is one of two
infectious diseases to have been eradicated, the other being
rinderpest, which was declared eradicated in 2011.
[16][17][18]
Classification
There were two clinical forms of smallpox. Variola major was the
severe and most common form, with a more extensive rash and higher
fever.
Variola minor was a less common presentation, and a much less severe disease, with historical death rates of 1% or less.
[19] Subclinical (
asymptomatic) infections with variola virus were noted but were not common.
[20] In addition, a form called
variola sine eruptione
(smallpox without rash) was seen generally in vaccinated persons. This
form was marked by a fever that occurred after the usual incubation
period and could be confirmed only by antibody studies or, rarely, by
virus isolation.
[20]
Signs and symptoms

Child showing rash due to ordinary-type smallpox (variola major)
The
incubation period
between contraction and the first obvious symptoms of the disease is
around 12 days. Once inhaled, variola major virus invades the
oropharyngeal (mouth and throat) or the
respiratory mucosa, migrates to regional
lymph nodes, and begins to multiply. In the initial growth phase the virus seems to move from cell to cell, but around the 12th day,
lysis of many infected cells occurs and the virus is found in the
bloodstream in large numbers (this is called
viremia), and a second wave of multiplication occurs in the spleen,
bone marrow, and lymph nodes. The initial or prodromal symptoms are similar to other viral diseases such as
influenza and the
common cold:
fever of at least 38.5 °C (101 °F),
muscle pain, malaise, headache and
prostration. As the
digestive tract
is commonly involved, nausea and vomiting and backache often occur. The
prodrome, or preeruptive stage, usually lasts 2–4 days. By days 12–15
the first visible lesions—small reddish spots called
enanthem—appear on mucous membranes of the mouth, tongue,
palate,
and throat, and temperature falls to near normal. These lesions rapidly
enlarge and rupture, releasing large amounts of virus into the
saliva.
[7]
Smallpox virus preferentially attacks skin cells, causing the characteristic pimples (called
macules)
associated with the disease. A rash develops on the skin 24 to 48 hours
after lesions on the mucous membranes appear. Typically the macules
first appear on the forehead, then rapidly spread to the whole face,
proximal portions of extremities, the trunk, and lastly to distal
portions of extremities. The process takes no more than 24 to 36 hours,
after which no new lesions appear.
[7]
At this point variola major infection can take several very different
courses, resulting in four types of smallpox disease based on the Rao
classification:
[21] ordinary, modified, malignant (or flat), and hemorrhagic. Historically, smallpox has an overall
fatality rate of about 30%; however, the malignant and hemorrhagic forms are usually fatal.
[22]
Ordinary
Ninety percent or more of smallpox cases among unvaccinated persons are of the ordinary type.
[20] In this form of the disease, by the second day of the rash the macules become raised
papules. By the third or fourth day the papules fill with an opalescent fluid to become
vesicles. This fluid becomes
opaque and
turbid within 24–48 hours, giving them the appearance of
pustules; however, the so-called pustules are filled with tissue debris, not pus.
[7]
By the sixth or seventh day, all the skin lesions have become
pustules. Between seven and ten days the pustules mature and reach their
maximum size. The pustules are sharply raised, typically round, tense,
and firm to the touch. The pustules are deeply embedded in the dermis,
giving them the feel of a small bead in the skin. Fluid slowly leaks
from the pustules, and by the end of the second week the pustules
deflate, and start to dry up, forming crusts (or scabs). By day 16–20
scabs have formed over all the lesions, which have started to flake off,
leaving
depigmented scars.
[23]
Ordinary smallpox generally produces a discrete rash, in which the
pustules stand out on the skin separately. The distribution of the rash
is densest on the face; denser on the extremities than on the trunk; and
on the extremities, denser on the distal parts than on the proximal.
The palms of the hands and soles of the feet are involved in the
majority of cases. Sometimes, the blisters merge into sheets, forming a
confluent rash, which begin to detach the outer layers of skin from the
underlying flesh. Patients with confluent smallpox often remain ill even
after scabs have formed over all the lesions. In one case series, the
case-fatality rate in confluent smallpox was 62%.
[20]

Man suffering from severe hemorrhagic-type smallpox.
Modified
Referring to the character of the eruption and the rapidity of its
development, modified smallpox occurs mostly in previously vaccinated
people. In this form the prodromal illness still occurs but may be less
severe than in the ordinary type. There is usually no fever during
evolution of the rash. The skin lesions tend to be fewer and evolve more
quickly, are more superficial, and may not show the uniform
characteristic of more typical smallpox.
[23] Modified smallpox is rarely, if ever, fatal. This form of variola major is more easily confused with
chickenpox.
[20]
Malignant
In malignant-type smallpox (also called flat smallpox) the lesions
remain almost flush with the skin at the time when raised vesicles form
in the ordinary type. It is unknown why some people develop this type.
Historically, it accounted for 5%–10% of cases, and the majority (72%)
were children.
[24] Malignant smallpox is accompanied by a severe
prodromal phase that lasts 3–4 days, prolonged high fever, and severe symptoms of
toxemia.
The rash on the tongue and palate is extensive. Skin lesions mature
slowly and by the seventh or eighth day they are flat and appear to be
buried in the skin. Unlike ordinary-type smallpox, the vesicles contain
little fluid, are soft and velvety to the touch, and may contain
hemorrhages. Malignant smallpox is nearly always fatal.
[20]
Hemorrhagic
Hemorrhagic smallpox is a severe form that is accompanied by extensive
bleeding into
the skin, mucous membranes, and gastrointestinal tract. This form
develops in approximately 2% of infections and occurred mostly in
adults.
[20]
In hemorrhagic smallpox the skin does not blister, but remains smooth.
Instead, bleeding occurs under the skin, making it look charred and
black,
[20] hence this form of the disease is also known as
black pox.
[25]
In the early, or fulminating form, hemorrhaging appears on the second or third day as sub-
conjunctival bleeding turns the whites of the eyes deep red. Hemorrhagic smallpox also produces a dusky
erythema,
petechiae, and hemorrhages in the spleen, kidney,
serosa, muscle, and, rarely, the
epicardium,
liver,
testes,
ovaries and
bladder.
Death often occurs suddenly between the fifth and seventh days of
illness, when only a few insignificant skin lesions are present. A later
form of the disease occurs in patients who survive for 8–10 days. The
hemorrhages appear in the early eruptive period, and the rash is flat
and does not progress beyond the vesicular stage.
[20] Patients in the early stage of disease show a decrease in
coagulation factors (e.g.
platelets,
prothrombin, and
globulin) and an increase in circulating
antithrombin. Patients in the late stage have significant
thrombocytopenia; however, deficiency of coagulation factors is less severe. Some in the late stage also show increased antithrombin.
[7] This form of smallpox occurs in anywhere from 3 to 25% of fatal cases depending on the virulence of the smallpox strain.
[22] Hemorrhagic smallpox is usually fatal.
[20]
Cause
Smallpox is caused by infection with variola virus, which belongs to the genus
Orthopoxvirus, the family
Poxviridae and subfamily chordopoxvirinae. Variola is a large brick-shaped virus measuring approximately 302 to 350
nanometers by 244 to 270 nm,
[26] with a single linear
double stranded DNA genome 186
kilobase pairs (kbp) in size and containing a
hairpin loop at each end.
[27][28] The two classic varieties of smallpox are variola major and variola minor.
Four orthopoxviruses cause infection in humans: variola,
vaccinia,
cowpox, and
monkeypox.
Variola virus infects only humans in nature, although primates and
other animals have been infected in a laboratory setting. Vaccinia,
cowpox, and monkeypox viruses can infect both humans and other animals
in nature.
[20]
The lifecycle of poxviruses is complicated by having multiple
infectious forms, with differing mechanisms of cell entry. Poxviruses
are unique among DNA viruses in that they replicate in the
cytoplasm of the cell rather than in the
nucleus. In order to replicate, poxviruses produce a variety of specialized proteins not produced by other
DNA viruses, the most important of which is a viral-associated
DNA-dependent RNA polymerase.
Both
enveloped and unenveloped virions are infectious. The viral envelope is made of modified
Golgi membranes containing viral-specific polypeptides, including
hemagglutinin.
[27] Infection with either variola major or variola minor confers immunity against the other.
[7]
Transmission
Transmission occurs through inhalation of
airborne variola virus, usually droplets expressed from the oral, nasal, or
pharyngeal mucosa
of an infected person. It is transmitted from one person to another
primarily through prolonged face-to-face contact with an infected
person, usually within a distance of 6 feet (1.8 m), but can also be
spread through direct contact with infected
bodily fluids or contaminated objects (
fomites)
such as bedding or clothing. Rarely, smallpox has been spread by virus
carried in the air in enclosed settings such as buildings, buses, and
trains.
[19] The virus can cross the
placenta, but the incidence of
congenital smallpox is relatively low.
[7] Smallpox is not notably infectious in the
prodromal period and viral shedding is usually delayed until the appearance of the rash, which is often accompanied by
lesions
in the mouth and pharynx. The virus can be transmitted throughout the
course of the illness, but is most frequent during the first week of the
rash, when most of the skin lesions are intact.
[20]
Infectivity wanes in 7 to 10 days when scabs form over the lesions, but
the infected person is contagious until the last smallpox scab falls
off.
[29]
Smallpox is highly contagious, but generally spreads more slowly and
less widely than some other viral diseases, perhaps because transmission
requires close contact and occurs after the onset of the rash. The
overall rate of infection is also affected by the short duration of the
infectious stage. In
temperate
areas, the number of smallpox infections were highest during the winter
and spring. In tropical areas, seasonal variation was less evident and
the disease was present throughout the year.
[20] Age distribution of smallpox infections depends on
acquired immunity.
Vaccination immunity declines over time and is probably lost in all but the most recently vaccinated populations.
[7] Smallpox is not known to be transmitted by insects or animals and there is no
asymptomatic carrier state.
[20]
Diagnosis
The clinical definition of smallpox is an illness with acute onset of
fever greater than 101 °F (38.3 °C) followed by a rash characterized by
firm, deep seated vesicles or pustules in the same stage of development
without other apparent cause.
[20] If a clinical case is observed, smallpox is confirmed using laboratory tests.
Microscopically, poxviruses produce characteristic
cytoplasmic inclusions, the most important of which are known as
Guarnieri bodies, and are the sites of
viral replication.
Guarnieri bodies are readily identified in skin biopsies stained with
hematoxylin and eosin, and appear as pink blobs. They are found in
virtually all poxvirus infections but the absence of Guarnieri bodies
cannot be used to rule out smallpox.
[30] The diagnosis of an orthopoxvirus infection can also be made rapidly by
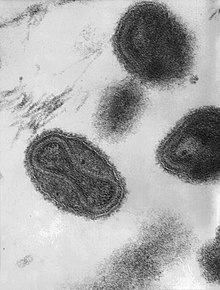
electron microscopic examination of pustular fluid or scabs. However, all orthopoxviruses exhibit identical brick-shaped
virions by electron microscopy.
[7]
Definitive laboratory identification of variola virus involves growing the virus on
chorioallantoic membrane (part of a chicken
embryo) and examining the resulting pock lesions under defined temperature conditions.
[31] Strains may be characterized by
polymerase chain reaction (PCR) and
restriction fragment length polymorphism (RFLP) analysis.
Serologic tests and
enzyme linked immunosorbent assays
(ELISA), which measure variola virus-specific immunoglobulin and
antigen have also been developed to assist in the diagnosis of
infection.
[32]
Chickenpox
was commonly confused with smallpox in the immediate post-eradication
era. Chickenpox and smallpox can be distinguished by several methods.
Unlike smallpox, chickenpox does not usually affect the palms and soles.
Additionally, chickenpox pustules are of varying size due to variations
in the timing of pustule eruption: smallpox pustules are all very
nearly the same size since the viral effect progresses more uniformly. A
variety of laboratory methods are available for detecting chickenpox in
evaluation of suspected smallpox cases.
[20]
-
-
In contrast to the rash in smallpox, the rash in
chickenpox occurs mostly on the torso, spreading less to the limbs.
Prevention
The earliest procedure used to prevent smallpox was
inoculation (also known as variolation). Inoculation was possibly practiced in India as early as 1000 BC,
[33] and involved either nasal
insufflation
of powdered smallpox scabs, or scratching material from a smallpox
lesion into the skin. However, the idea that inoculation originated in
India has been challenged as few of the ancient
Sanskrit medical texts described the process of inoculation.
[34]
Accounts of inoculation against smallpox in China can be found as early
as the late 10th century, and the procedure was widely practiced by the
16th century, during the
Ming Dynasty.
[35] If successful, inoculation produced lasting
immunity
to smallpox. However, because the person was infected with variola
virus, a severe infection could result, and the person could transmit
smallpox to others. Variolation had a 0.5–2% mortality rate,
considerably less than the 20–30% mortality rate of the disease itself.
[20]
Lady Mary Wortley Montagu observed smallpox inoculation during her stay in the
Ottoman Empire,
writing detailed accounts of the practice in her letters, and
enthusiastically promoted the procedure in England upon her return in
1718.
[36] In 1721,
Cotton Mather and colleagues provoked controversy in Boston by inoculating hundreds. In 1796,
Edward Jenner, a doctor in
Berkeley, Gloucestershire, rural England, discovered that immunity to smallpox could be produced by inoculating a person with material from a
cowpox lesion. Cowpox is a poxvirus in the same family as variola. Jenner called the material used for inoculation
vaccine, from the
root word vacca, which is
Latin
for cow. The procedure was much safer than variolation, and did not
involve a risk of smallpox transmission. Vaccination to prevent smallpox
was soon practiced all over the world. During the 19th century, the
cowpox virus used for smallpox vaccination was replaced by
vaccinia virus. Vaccinia is in the same family as cowpox and variola but is
genetically distinct from both. The origin of vaccinia virus and how it came to be in the vaccine are not known.
[20]

An 1802 cartoon of the early controversy surrounding
Edward Jenner's vaccination theory, showing using his
cowpox-derived smallpox vaccine causing cattle to emerge from patients.
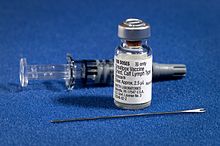
The current formulation of smallpox vaccine is a live virus
preparation of infectious vaccinia virus. The vaccine is given using a
bifurcated (two-pronged) needle that is dipped into the vaccine
solution. The needle is used to prick the skin (usually the upper arm) a
number of times in a few seconds. If successful, a red and itchy bump
develops at the vaccine site in three or four days. In the first week,
the bump becomes a large blister (called a "Jennerian vesicle") which
fills with pus, and begins to drain. During the second week, the blister
begins to dry up and a scab forms. The scab falls off in the third
week, leaving a small scar.
[37]
The
antibodies
induced by vaccinia vaccine are cross-protective for other
orthopoxviruses, such as monkeypox, cowpox, and variola (smallpox)
viruses. Neutralizing antibodies are detectable 10 days after first-time
vaccination, and seven days after revaccination. Historically, the
vaccine has been effective in preventing smallpox infection in 95% of
those vaccinated.
[38]
Smallpox vaccination provides a high level of immunity for three to
five years and decreasing immunity thereafter. If a person is vaccinated
again later, immunity lasts even longer. Studies of smallpox cases in
Europe in the 1950s and 1960s demonstrated that the fatality rate among
persons vaccinated less than 10 years before exposure was 1.3%; it was
7% among those vaccinated 11 to 20 years prior, and 11% among those
vaccinated 20 or more years prior to infection. By contrast, 52% of
unvaccinated persons died.
[39]

A demonstration by medical personnel on use of a bifurcated needle to deliver the smallpox vaccine, 2002.
There are side effects and risks associated with the smallpox
vaccine. In the past, about 1 out of 1,000 people vaccinated for the
first time experienced serious, but non-life-threatening, reactions
including toxic or
allergic reaction at the site of the vaccination (
erythema multiforme),
spread of the vaccinia virus to other parts of the body, and to other
individuals. Potentially life-threatening reactions occurred in 14 to
500 people out of every 1 million people vaccinated for the first time.
Based on past experience, it is estimated that 1 or 2 people in 1
million (0.000198%) who receive the vaccine may die as a result, most
often the result of postvaccinial
encephalitis or severe
necrosis in the area of vaccination (called progressive vaccinia).
[38]
Given these risks, as smallpox became effectively eradicated and the
number of naturally occurring cases fell below the number of
vaccine-induced illnesses and deaths, routine childhood vaccination was
discontinued in the United States in 1972, and was abandoned in most
European countries in the early 1970s.
[5][40]
Routine vaccination of health care workers was discontinued in the U.S.
in 1976, and among military recruits in 1990 (although military
personnel deploying to the Middle East and Korea still receive the
vaccination.
[41]) By 1986, routine vaccination had ceased in all countries.
[5] It is now primarily recommended for laboratory workers at risk for occupational exposure.
[20]
Treatment
Smallpox vaccination within three days of exposure will prevent or
significantly lessen the severity of smallpox symptoms in the vast
majority of people. Vaccination four to seven days after exposure can
offer some protection from disease or may modify the severity of
disease.
[38]
Other than vaccination, treatment of smallpox is primarily supportive,
such as wound care and infection control, fluid therapy, and possible
ventilator assistance. Flat and hemorrhagic types of smallpox are treated with the same therapies used to treat
shock, such as
fluid resuscitation.
People with semi-confluent and confluent types of smallpox may have
therapeutic issues similar to patients with extensive skin
burns.
[42]
No drug is currently approved for the treatment of smallpox. However,
antiviral treatments have improved since the last large smallpox epidemics, and studies suggest that the antiviral drug
cidofovir might be useful as a therapeutic agent. The drug must be administered
intravenously, however, and may cause serious
kidney toxicity.
[43]
Prognosis
The overall case-fatality rate for ordinary-type smallpox is about
30%, but varies by pock distribution: ordinary type-confluent is fatal
about 50–75% of the time, ordinary-type semi-confluent about 25–50% of
the time, in cases where the rash is discrete the case-fatality rate is
less than 10%. The overall fatality rate for children younger than 1
year of age is 40–50%. Hemorrhagic and flat types have the highest
fatality rates. The fatality rate for flat-type is 90% or greater and
nearly 100% is observed in cases of hemorrhagic smallpox. The
case-fatality rate for variola minor is 1% or less.
[23] There is no evidence of chronic or recurrent infection with variola virus.
[23]
In fatal cases of ordinary smallpox, death usually occurs between the
tenth and sixteenth days of the illness. The cause of death from
smallpox is not clear, but the infection is now known to involve
multiple organs. Circulating
immune complexes, overwhelming
viremia, or an uncontrolled
immune response may be contributing factors.
[20]
In early hemorrhagic smallpox, death occurs suddenly about six days
after the fever develops. Cause of death in hemorrhagic cases involved
heart failure, sometimes accompanied by
pulmonary edema. In late hemorrhagic cases, high and sustained viremia, severe
platelet loss and poor immune response were often cited as causes of death.
[24] In flat smallpox modes of death are similar to those in burns, with loss of fluid, protein and
electrolytes beyond the capacity of the body to replace or acquire, and fulminating
sepsis.
[42]
Complications
Complications of smallpox arise most commonly in the
respiratory system and range from simple
bronchitis to fatal
pneumonia.
Respiratory complications tend to develop on about the eighth day of
the illness and can be either viral or bacterial in origin. Secondary
bacterial infection of the skin is a relatively uncommon complication of smallpox. When this occurs, the fever usually remains elevated.
[20]
Other complications include
encephalitis
(1 in 500 patients), which is more common in adults and may cause
temporary disability; permanent pitted scars, most notably on the face;
and complications involving the eyes (2% of all cases). Pustules can
form on the eyelid,
conjunctiva, and
cornea, leading to complications such as
conjunctivitis,
keratitis,
corneal ulcer,
iritis,
iridocyclitis, and optic
atrophy.
Blindness
results in approximately 35% to 40% of eyes affected with keratitis and
corneal ulcer. Hemorrhagic smallpox can cause subconjunctival and
retinal hemorrhages. In 2% to 5% of young children with smallpox, virions reach the joints and bone, causing
osteomyelitis variolosa. Lesions are symmetrical, most common in the elbows,
tibia, and
fibula, and characteristically cause separation of an
epiphysis and marked
periosteal reactions. Swollen joints limit movement, and
arthritis may lead to limb deformities,
ankylosis, malformed bones, flail joints, and stubby fingers.
[7]
History
Viral evolution
The date of the appearance of smallpox is not settled. It most likely
evolved from a rodent virus between 68,000 and 16,000 years ago.
[44][45] The wide range of dates is due to the different records used to calibrate the molecular clock. One
clade
was the variola major strains (the more clinically severe form of
smallpox) which spread from Asia between 400 and 1,600 years ago. A
second clade included both alastrim minor (a phenotypically mild
smallpox) described from the American continents and isolates from West
Africa which diverged from an ancestral strain between 1,400 and 6,300
years before present. This clade further diverged into two subclades at
least 800 years ago.
A second estimate has placed the separation of variola from
Taterapox at 3000–4000 years ago.
[46]
This is consistent with archaeological and historical evidence
regarding the appearance of smallpox as a human disease which suggests a
relatively recent origin. However if the mutation rate is assumed to be
similar to that of the
herpesviruses the divergence date between variola from
Taterapox has been estimated to be 50,000 years ago.
[46]
While this is consistent with the other published estimates it suggests
that the archaeological and historical evidence is very incomplete.
Better estimates of mutation rates in these viruses are needed.
It seems to have emerged in its endemic form in
India 2500–3000 years ago.
[47]
Other history
The earliest credible clinical evidence of smallpox is found in the
smallpox-like disease in medical writings from ancient India (as early
as 1500 BC),
[48] Egyptian mummy of
Ramses V who died more than 3000 years ago (1145 BC).
[9] and China (1122 BC).
[49]
It has been speculated that Egyptian traders brought smallpox to India
during the 1st millennium BC, where it remained as an endemic human
disease for at least 2000 years. Smallpox was probably introduced into
China during the 1st century AD from the southwest, and in the 6th
century was carried from China to Japan.
[24] In Japan, the epidemic of 735–737 is believed to have killed as much as one-third of the population.
[10][50] At least seven religious deities have been specifically dedicated to smallpox, such as the god
Sopona in the
Yoruba religion. In India, the Hindu goddess of smallpox,
Sitala Mata, was worshiped in temples throughout the country.
[51]

Statue of
Sopona, the West African god thought to inflict the disease.
The timing of the arrival of smallpox in Europe and south-western
Asia is less clear. Smallpox is not clearly described in either the
Old or
New Testaments of the Bible or in the literature of the Greeks or Romans. While some have identified the
Plague of Athens – which was said to have originated in "
Ethiopia" and Egypt – or the plague that lifted Carthage's 396 BC
siege of Syracuse with smallpox,
[2] many scholars agree it is very unlikely such a serious disease as variola major would have escaped being described by
Hippocrates if it had existed in the Mediterranean region during his lifetime.
[52] While the
Antonine Plague that swept through the
Roman Empire in AD 165–180 may have been caused by smallpox,
[53] Saint
Nicasius of Rheims became the patron saint of smallpox victims for having supposedly survived a bout in 450,
[2] and Saint
Gregory of Tours recorded a similar outbreak in France and Italy in 580, the first use of the term
variola;
[2] other historians speculate that
Arab armies first carried smallpox from Africa into Southwestern Europe during the 7th and 8th centuries.
[24] In the 9th century the
Persian physician,
Rhazes, provided one of the most definitive descriptions of smallpox and was the first to differentiate smallpox from
measles and
chickenpox in his
Kitab fi al-jadari wa-al-hasbah (
The Book of Smallpox and Measles).
[54] During the
Middle Ages,
smallpox made periodic incursions into Europe but did not become
established there until the population increased and population movement
became more active during the era of the
Crusades. By the 16th century smallpox had become well established across most of Europe.
[24]
With its introduction into populated areas in India, China and Europe,
smallpox affected mainly children, with periodic epidemics that killed
as many as 30% of those infected. The settled existence of smallpox in
Europe was of particular historical importance, since successive waves
of exploration and colonization by Europeans tended to spread the
disease to other parts of the world. By the 16th century it had become
an important cause of morbidity and mortality throughout much of the
world.
[24]

Drawing accompanying text in Book XII of the 16th-century
Florentine Codex (compiled 1540–1585), showing
Nahuas of conquest-era central Mexico suffering from smallpox.
There are no credible descriptions of smallpox-like disease in the
Americas before the westward exploration by Europeans in the 15th century AD.
[45] Smallpox was introduced into the Caribbean island of
Hispaniola
in 1509, and into the mainland in 1520, when Spanish settlers from
Hispaniola arrived in Mexico bringing smallpox with them. Smallpox
devastated the native
Amerindian population and was an important factor in the conquest of the
Aztecs and the
Incas by the Spaniards.
[24] Settlement of the east coast of North America in 1633 in
Plymouth, Massachusetts was also accompanied by devastating outbreaks of smallpox among Native American populations,
[55] and subsequently among the native-born colonists.
[56] Some estimates indicate case fatality rates of 80–90% in Native American populations during smallpox epidemics.
[57] Smallpox was introduced into
Australia in 1789 and again in 1829.
[24] Although the disease was never endemic on the continent,
[24] it was the principal cause of death in
Aboriginal populations between 1780 and 1870.
[58]

Smallpox sufferer in the United States, 1912
By the mid-18th century smallpox was a major
endemic disease
everywhere in the world except in Australia and in several small
islands. In Europe smallpox was a leading cause of death in the 18th
century, killing an estimated 400,000 Europeans each year.
[59] Through the century smallpox resulted in the deaths of perhaps 10% of all the infants of
Sweden every year,
[10] and the death rate of infants in
Russia may have been even higher.
[49] The widespread use of
variolation
in a few countries, notably Great Britain, its North American colonies,
and China, somewhat reduced the impact of smallpox among the wealthy
classes during the latter part of the 18th century, but a real reduction
in its incidence did not occur until vaccination became a common
practice toward the end of the 19th century. Improved vaccines and the
practice of re-vaccination led to a substantial reduction in cases in
Europe and North America, but smallpox remained almost unchecked
everywhere else in the world. In the United States and South Africa a
much milder form of smallpox,
variola minor, was recognized just before the close of the 19th century. By the mid-20th century
variola minor occurred along with
variola major, in varying proportions, in many parts of Africa. Patients with
variola minor experience only a mild systemic illness, are often
ambulant throughout the course of the disease, and are therefore able to more easily spread disease. Infection with
v. minor induces immunity against the more deadly
variola major form. Thus as
v. minor
spread all over the USA, into Canada, the South American countries and
Great Britain it became the dominant form of smallpox, further reducing
mortality rates.
[24]
Eradication

Vaccination during the Smallpox Eradication and Measles Control Program in
Niger, February, 1969.
The English physician
Edward Jenner
demonstrated the effectiveness of cowpox to protect humans from
smallpox in 1796, after which various attempts were made to eliminate
smallpox on a regional scale. As early as 1803, the Spanish Crown
organized a mission (the
Balmis expedition) to transport the vaccine to the
Spanish colonies in the Americas and the Philippines, and establish mass vaccination programs there.
[60] The
U.S. Congress passed the
Vaccine Act of 1813
to ensure that safe smallpox vaccine would be available to the American
public. By about 1817, a very solid state vaccination program existed
in the
Dutch East Indies.
[61] In
British India
a program was launched to propagate smallpox vaccination, through
Indian vaccinators, under the supervision of European officials.
[62]
Nevertheless, British vaccination efforts in India, and in Burma in
particular, were hampered by stubborn indigenous preference for
inoculation and distrust of vaccination, despite tough legislation,
improvements in the local efficacy of the vaccine and vaccine
preservative, and education efforts.
[63] By 1832, the federal government of the United States established a smallpox vaccination program for
Native Americans.
[64] In 1842, the United Kingdom banned inoculation, later progressing to
mandatory vaccination. The British government introduced compulsory smallpox vaccination by an Act of Parliament in 1853.
[65]
In the United States, from 1843 to 1855 first Massachusetts, and then
other states required smallpox vaccination. Although some disliked these
measures,
[49]
coordinated efforts against smallpox went on, and the disease continued
to diminish in the wealthy countries. By 1897, smallpox had largely
been eliminated from the United States.
[66]
In Northern Europe a number of countries had eliminated smallpox by
1900, and by 1914, the incidence in most industrialized countries had
decreased to comparatively low levels. Vaccination continued in
industrialized countries, until the mid to late 1970s as protection
against reintroduction. Australia and New Zealand are two notable
exceptions; neither experienced endemic smallpox and never vaccinated
widely, relying instead on protection by distance and strict
quarantines.
[67]

Smallpox quarantine order, California, ca 1910
The first
hemisphere-wide effort to eradicate smallpox was made in 1950 by the
Pan American Health Organization.
[68]
The campaign was successful in eliminating smallpox from all American
countries except Argentina, Brazil, Colombia, and Ecuador.
[67] In 1958 Professor
Viktor Zhdanov, Deputy Minister of Health for the
USSR, called on the
World Health Assembly to undertake a global initiative to
eradicate smallpox. The proposal (Resolution WHA11.54) was accepted in 1959.
[69]
At this point, 2 million people were dying from smallpox every year.
Overall, however, the progress towards eradication was disappointing,
especially in Africa and in the
Indian subcontinent. In 1966 an international team, the Smallpox Eradication Unit, was formed under the leadership of an American,
Donald Henderson.
[70]
In 1967, the World Health Organization intensified the global smallpox
eradication by contributing $2.4 million annually to the effort, and
adopted the new
disease surveillance method promoted by Czech epidemiologist
Karel Raška.
[71][72]

Two-year old
Rahima Banu of Bangladesh (pictured) was the last person infected with naturally occurring
Variola major, in 1975
In the early 1950s an estimated 50 million cases of smallpox occurred in the world each year.
[5]
To eradicate smallpox, each outbreak had to be stopped from spreading,
by isolation of cases and vaccination of everyone who lived close by.
This process is known as "ring vaccination". The key to this strategy
was monitoring of cases in a community (known as surveillance) and
containment. The initial problem the WHO team faced was inadequate
reporting of smallpox cases, as many cases did not come to the attention
of the authorities. The fact that humans are the only reservoir for
smallpox infection, and that
carriers
did not exist, played a significant role in the eradication of
smallpox. The WHO established a network of consultants who assisted
countries in setting up surveillance and containment activities. Early
on donations of vaccine were provided primarily by the Soviet Union and
the United States, but by 1973, more than 80% of all vaccine was
produced in developing countries.
[67]
The last major European outbreak of smallpox was in
1972 in Yugoslavia, after a pilgrim from
Kosovo
returned from the Middle East, where he had contracted the virus. The
epidemic infected 175 people, causing 35 deaths. Authorities declared
martial law,
enforced quarantine, and undertook widespread re-vaccination of the
population, enlisting the help of the WHO. In two months, the outbreak
was over.
[73] Prior to this, there had been a smallpox outbreak in May–July 1963 in
Stockholm, Sweden, brought from the
Far East by a Swedish sailor; this had been dealt with by quarantine measures and vaccination of the local population.
[74]
By the end of 1975, smallpox persisted only in the
Horn of Africa.
Conditions were very difficult in Ethiopia and Somalia, where there
were few roads. Civil war, famine, and refugees made the task even more
difficult. An intensive surveillance and containment and vaccination
program was undertaken in these countries in early and mid-1977, under
the direction of Australian microbiologist
Frank Fenner. As the campaign neared its goal, Fenner and his team played an important role in verifying eradication.
[75] The last naturally occurring case of indigenous smallpox (
Variola minor) was diagnosed in
Ali Maow Maalin, a hospital cook in Merca,
Somalia, on 26 October 1977.
[20] The last naturally occurring case of the more deadly
Variola major had been detected in October 1975 in a two-year-old
Bangladeshi girl,
Rahima Banu.
[25]
The global eradication of smallpox was certified, based on intense
verification activities in countries, by a commission of eminent
scientists on 9 December 1979 and subsequently endorsed by the World
Health Assembly on 8 May 1980.
[5][76] The first two sentences of the resolution read:
"Having considered the development and results of the global program
on smallpox eradication initiated by WHO in 1958 and intensified since
1967 … Declares solemnly that the world and its peoples have won freedom
from smallpox, which was a most devastating disease sweeping in
epidemic form through many countries since earliest time, leaving death,
blindness and disfigurement in its wake and which only a decade ago was
rampant in Africa, Asia and South America."
—World Health Organization, Resolution WHA33.3
[77]
Post-eradication

Three former directors of the Global Smallpox Eradication Program read the news that smallpox had been globally eradicated, 1980
The last cases of smallpox in the world occurred in an outbreak of two cases (one of which was fatal) in
Birmingham,
UK in 1978. A medical photographer,
Janet Parker, contracted the disease at the
University of Birmingham Medical School and died on September 11, 1978,
[77] after which the scientist responsible for smallpox research at the university, Professor Henry Bedson, committed
suicide.
[4]
In light of this incident, all known stocks of smallpox were destroyed
or transferred to one of two WHO reference laboratories which had
BSL-4 facilities; the
Centers for Disease Control and Prevention (CDC) in the United States and the
State Research Center of Virology and Biotechnology VECTOR in
Koltsovo, Russia.
[78]
In 1986, the
World Health Organization
first recommended destruction of the virus, and later set the date of
destruction to be 30 December 1993. This was postponed to 30 June 1999.
[79]
Due to resistance from the U.S. and Russia, in 2002 the World Health
Assembly agreed to permit the temporary retention of the virus stocks
for specific research purposes.
[80]
Destroying existing stocks would reduce the risk involved with ongoing
smallpox research; the stocks are not needed to respond to a smallpox
outbreak.
[81] Some scientists have argued that the stocks may be useful in developing new vaccines, antiviral drugs, and diagnostic tests;
[82] however, a 2010 review by a team of public health experts appointed by the
World Health Organization concluded that no essential public health purpose is served by the U.S. and Russia continuing to retain virus stocks.
[83]
The latter view is frequently supported in the scientific community,
particularly among veterans of the WHO Smallpox Eradication Program.
[84]
In March 2004 smallpox
scabs were found tucked inside an envelope in a book on
Civil War medicine in
Santa Fe, New Mexico.
[85] The envelope was labeled as containing scabs from a vaccination and gave scientists at the
Centers for Disease Control and Prevention an opportunity to study the history of smallpox vaccination in the U.S.
Society and culture
Biological warfare
The British considered using smallpox as a
biological warfare agent at the
Siege of Fort Pitt during the
French and Indian Wars (1754–63) against France and its
Native American allies.
[86][87]
Although it is not clear whether the actual use of smallpox had
official sanction, on June 24, 1763, William Trent, a local trader,
wrote, "Out of our regard for them [sc. representatives of the besieging
Delawares], we gave them two Blankets and an Handkerchief out of the
Small Pox Hospital. I hope it will have the desired effect."
[88][89]
Historians do not agree on whether this effort to broadcast the disease
was successful. It has also been alleged that smallpox was used as a
weapon during the
American Revolutionary War (1775–83).
[90][91]
Subsequently, according to an article in
Journal of Australian Studies, in 1789, British marines used smallpox against indigenous tribes in New South Wales.
[92] This occasion was also discussed earlier in
Bulletin of the History of Medicine[93] and by David Day in his book
Claiming a Continent A New History of Australia.
[94]
During
World War II, scientists from the United Kingdom, United States and Japan (
Unit 731 of the imperial Japanese army) were involved in research into producing a biological weapon from smallpox.
[95]
Plans of large scale production were never carried through as they
considered that the weapon would not be very effective due to the
wide-scale availability of a
vaccine.
[96]
In 1947 the
Soviet Union established a smallpox weapons factory in the city of
Zagorsk, 75 km to the northeast of Moscow.
[97] An
outbreak of weaponized smallpox occurred during testing at a facility on an island in the
Aral Sea in 1971. General Prof. Peter Burgasov, former Chief Sanitary Physician of the
Soviet Army and a senior researcher within the
Soviet program of biological weapons, described the incident:
On Vozrozhdeniya Island in the Aral Sea, the strongest recipes of smallpox were tested. Suddenly I was informed that there were mysterious cases of mortalities in Aralsk.
A research ship of the Aral fleet came to within 15 km of the island
(it was forbidden to come any closer than 40 km). The lab technician of
this ship took samples of plankton twice a day from the top deck. The
smallpox formulation—400 gr. of which was exploded on the island—"got
her" and she became infected. After returning home to Aralsk, she
infected several people including children. All of them died. I
suspected the reason for this and called the Chief of General Staff of
Ministry of Defense and requested to forbid the stop of the Alma-Ata—Moscow train in Aralsk. As a result, the epidemic around the country was prevented. I called Andropov, who at that time was Chief of KGB, and informed him of the exclusive recipe of smallpox obtained on Vozrazhdenie Island.[98][99]
Others contend that the first patient may have contracted the disease while visiting Uyaly or
Komsomolsk-on-Ustyurt, two cities where the boat docked.
[100][101]
Responding to international pressures, in 1991 the Soviet government
allowed a joint U.S.-British inspection team to tour four of its main
weapons facilities at
Biopreparat. The inspectors were met with evasion and denials from the Soviet scientists, and were eventually ordered out of the facility.
[102] In 1992 Soviet defector
Ken Alibek
alleged that the Soviet bioweapons program at Zagorsk had produced a
large stockpile—as much as twenty tons—of weaponized smallpox (possibly
engineered to resist vaccines, Alibek further alleged), along with
refrigerated
warheads to deliver it. Alibek's stories about the former Soviet program's smallpox activities have never been independently verified.
In 1997, the Russian government announced that all of its remaining smallpox samples would be moved to the
Vector Institute in
Koltsovo.
[102]
With the breakup of the Soviet Union and unemployment of many of the
weapons program's scientists, U.S. government officials have expressed
concern that smallpox and the expertise to weaponize it may have become
available to other governments or terrorist groups who might wish to use
virus as means of biological warfare.
[103] Specific allegations made against Iraq in this respect, however, proved to be false.
[104]
Concern has been expressed by some that
artificial gene synthesis could be used to recreate the virus from existing digital genomes, for use in biological warfare.
[105] Insertion of the synthesized smallpox DNA into existing related
pox viruses could theoretically be used to recreate the virus.
[105]
The first step to mitigating this risk, it has been suggested, should
be to destroy the remaining virus stocks so as to enable unequivocal
criminalization of any possession of the virus.
[106]
Notable cases
Famous historical figures who contracted smallpox include Lakota Chief
Sitting Bull,
Ramses V of
Egypt,
[107] the
Kangxi Emperor (survived),
Shunzhi Emperor and
Tongzhi Emperor (refer to the official history) of China,
Date Masamune of Japan (who lost an eye to the disease).
Cuitláhuac, the 10th
tlatoani (ruler) of the
Aztec city of
Tenochtitlan, died of smallpox in 1520, shortly after its introduction to the
Americas, and the Incan emperor
Huayna Capac died of it in 1527. More recent public figures include
Guru Har Krishan, 8th Guru of the Sikhs, in 1664,
Peter II of Russia in 1730 (died),
[108] George Washington (survived), king
Louis XV in 1774 (died) and
Maximilian III Joseph, Elector of Bavaria in 1777.
Prominent families throughout the world often had several people
infected by and/or perish from the disease. For example, several
relatives of
Henry VIII survived the disease but were scarred by it. These include his sister
Margaret, Queen of Scotland, his fourth wife,
Anne of Cleves, and his two daughters:
Mary I of England in 1527 and
Elizabeth I of England in 1562 (as an adult she would often try to disguise the pockmarks with heavy makeup). His great-niece,
Mary, Queen of Scots, contracted the disease as a child but had no visible scarring.
In Europe, deaths from smallpox often changed dynastic succession. The only surviving son of
Henry VIII,
Edward VI,
died from complications shortly after apparently recovering from the
disease, thereby nullifying Henry's efforts to ensure a male successor
to the throne (his immediate successors were all females).
Louis XV of France succeeded his great-grandfather
Louis XIV
through a series of deaths of smallpox or measles among those earlier
in the succession line. He himself died of the disease in 1774.
William III lost his mother to the disease when he was only ten years old in 1660, and named his uncle
Charles
as legal guardian: her death from smallpox would indirectly spark a
chain of events that would eventually lead to the permanent ousting of
the Stuart line from the British throne. William III's wife,
Mary II of England, died from smallpox as well.
In Russia,
Peter II of Russia died of the disease at 15 years of age. Also, prior to becoming
Russian Emperor,
Peter III caught the virus and suffered greatly from it. He was left scarred and disfigured. His wife,
Catherine the Great, was spared but fear of the virus clearly had its effects on her. She feared for her son and heir
Pavel's
safety so much that she made sure that large crowds were kept at bay
and sought to isolate him. Eventually, she decided to have herself
inoculated by a
Scottish
doctor, Thomas Dimsdale. While this was considered a controversial
method at the time, she succeeded. Her son Pavel was later inoculated as
well. Catherine then sought to have inoculations throughout her empire
stating: "My objective was, through my example, to save from death the
multitude of my subjects who, not knowing the value of this technique,
and frightened of it, were left in danger." By 1800, approximately 2
million inoculations were administered in the Russian Empire.
[109]
In China, the
Qing Dynasty had extensive protocols to protect
Manchus from
Peking's endemic smallpox.
U.S. Presidents
George Washington,
Andrew Jackson, and
Abraham Lincoln all contracted and recovered from the disease. Washington became infected with smallpox on a visit to
Barbados in 1751.
[110]
Jackson developed the illness after being taken prisoner by the British
during the American Revolution, and though he recovered, his brother
Robert did not.
[110]
Lincoln contracted the disease during his Presidency, possibly from his
son Tad, and was quarantined shortly after giving the Gettysburg
address in 1863.
[110]
Famous theologian
Jonathan Edwards died of smallpox in 1758 following an inoculation.
[111]
Soviet leader
Joseph Stalin
fell ill with smallpox at the age of seven. His face was badly scarred
by the disease. He later had photographs retouched to make his pockmarks
less apparent.
[112]
Hungarian poet
Ferenc Kölcsey, who wrote the Hungarian national anthem, lost his right eye to smallpox.
[113]
Tradition and religion

The Hindu goddess
Shitala was worshipped to prevent or cure smallpox.
As a reaction to the devastation of smallpox, smallpox gods and
goddesses were invented as a mechanism to cope with the disease. Two
examples of this occurred in China and India. In China, the smallpox
goddess was referred to as T’ou-Shen Niang-Niang.
[114]
The Chinese actively worked to please the goddess and thus keep the
disease at bay. For example, the Chinese referred to the smallpox
pustules as "beautiful flowers"; this was an attempt to not offend the
goddess and keep her happy.
[115]
The Chinese also took great measure to protect children from the
dangers of smallpox by tricking their smallpox goddess. It was believed
that the goddess enjoyed passing the disease to attractive children.
This transmission was most likely to occur on the last night of the
year, so children wore ugly masks to bed to trick the goddess into
passing over them.
[115]
If infection of smallpox did occur, shrines were set up in the homes of
the victims. These shrines were worshipped and offerings made to while
the victim was sick. If the victim recovered, the shrines were taken
away from the home in a special handmade paper chair or boat and burned.
If the patient did not recover, the shrine was destroyed and curses
were used to remove the goddess from the house.
[114]
India’s first records of smallpox can be found in a medical book that
dates back to A.D. 400. This book describes a disease that sounds
exceptionally like smallpox.
[115] India, like China, created a goddess in response to its exposure to smallpox. The Hindu goddess
Shitala
was both worshipped and feared during her reign. It was believed that
this goddess was both evil and kind and had the ability to inflict
victims when angered, as well as calm the fevers of the already
afflicted.
[116]
Portraits of the goddess show her holding a broom in her right hand to
continue to move the disease and a pot of cool water in the other hand
in an attempt to soothe victims.
[115]
Shrines were created where many India natives, both healthy and not,
went to worship and attempt to protect themselves from this disease.
Some Indian women, in an attempt to ward off Shitala, placed plates of
cooling foods and pots of water on the roofs of their homes.
[117]
In medieval times, several countries held a belief in the
smallpox demon,
who was blamed for the disease. Such beliefs were prominent in Japan,
Europe, Africa, and other parts of the world. Nearly all cultures who
believed in the demon also believed that it was afraid of the color red.
This led to the
red treatment, where victims and their rooms
would be decorated in red. The practice spread to Europe in the 12th
century and was practiced by (among others)
Charles V of France and
Elizabeth I of England.
[2] Given scientific credibility by some studies by Finsen showing red light reduced scarring,
[2] the belief persisted until the 1930s.
See also